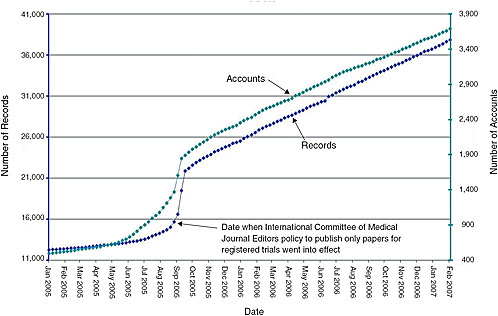
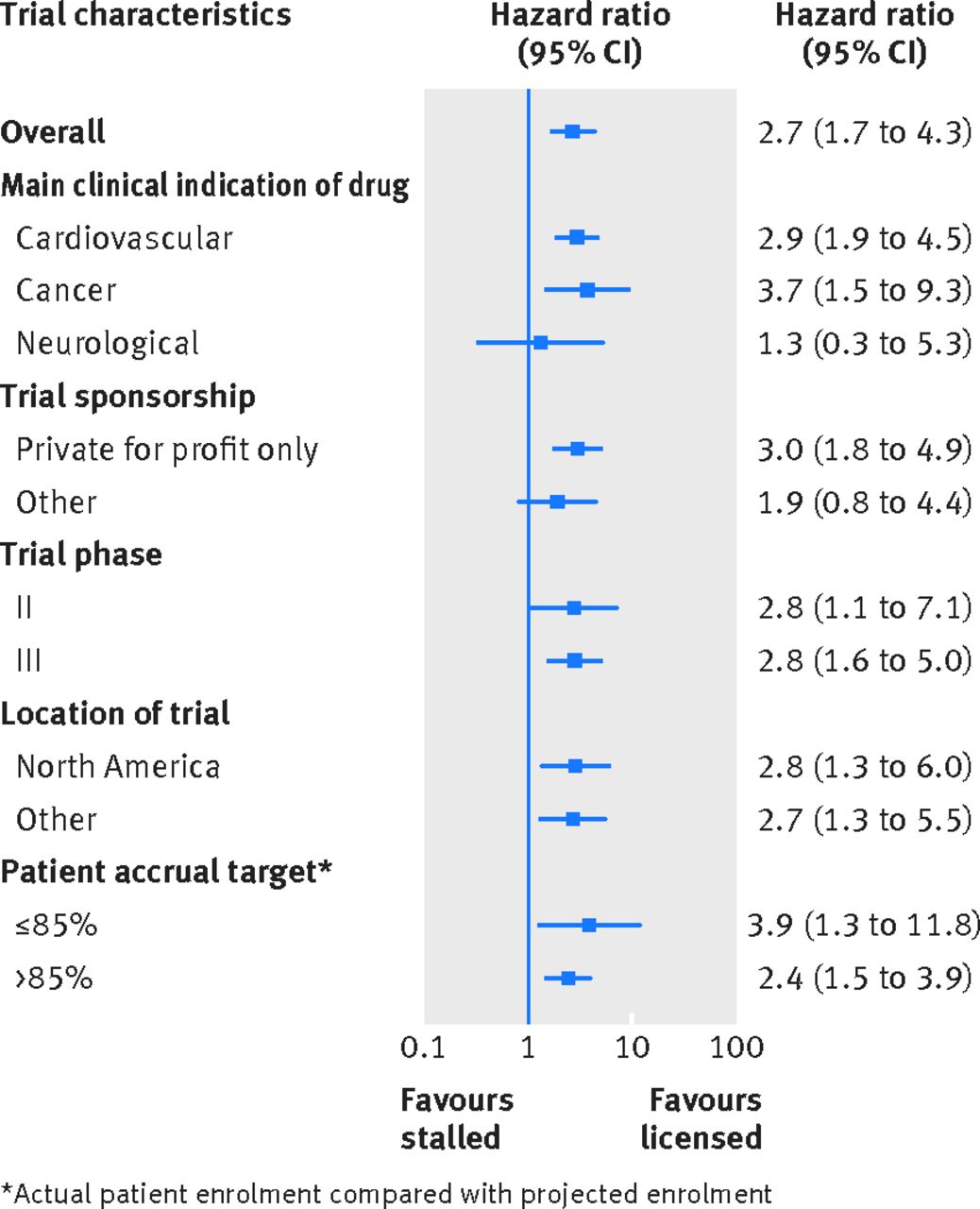
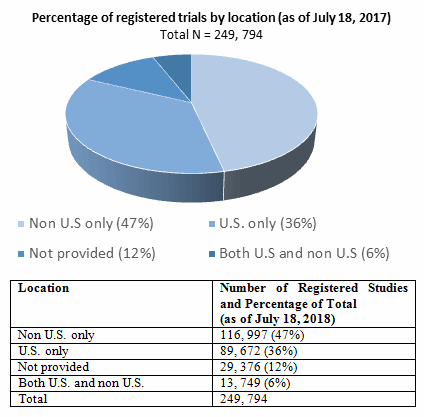
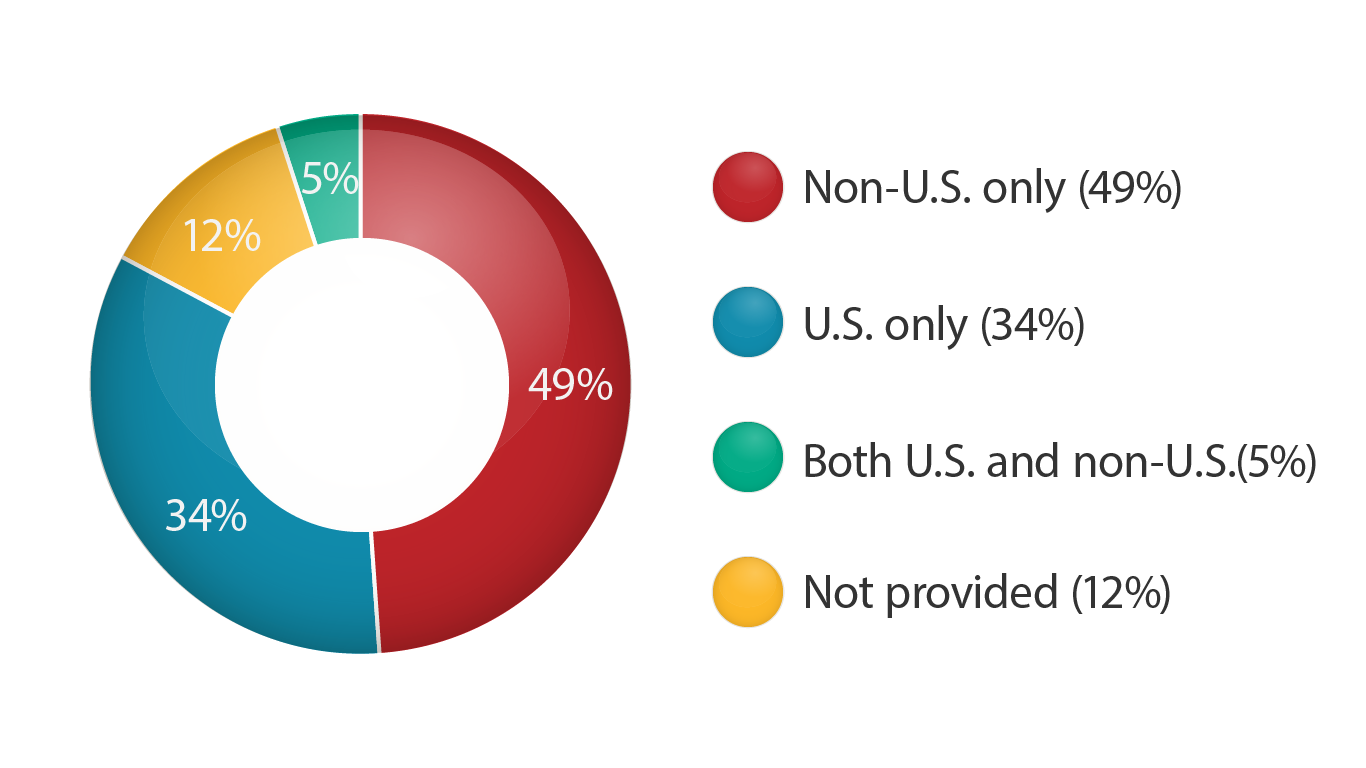
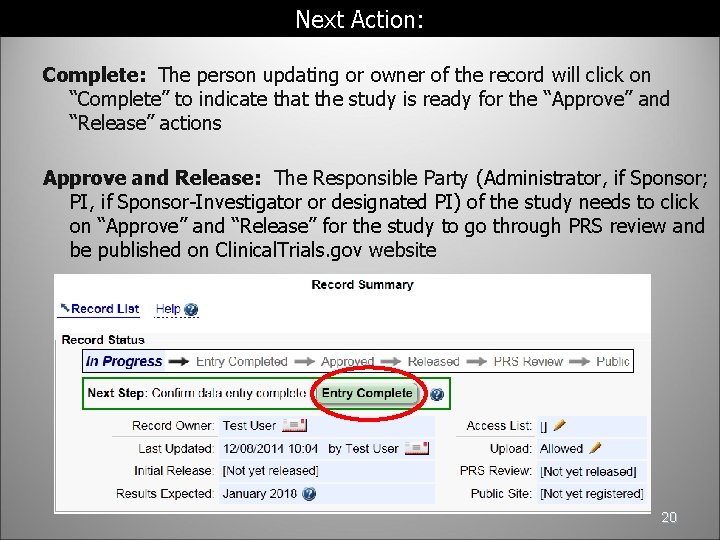
Regulatory Approval – Clinical Trial Medical Monitoring Plan | Online Clinical Research Courses In India

Saket Gokhale on Twitter: "Phase 3 of Covaxin trials were registered on 9th November, 2020 with a sample size of 25,800 participants. Registered duration for Phase 3 is 1 year i.e. by

A group of parents are officially lobbying to register their children as part of a 'control group' for the Covid-19 vaccine. : r/insaneparents
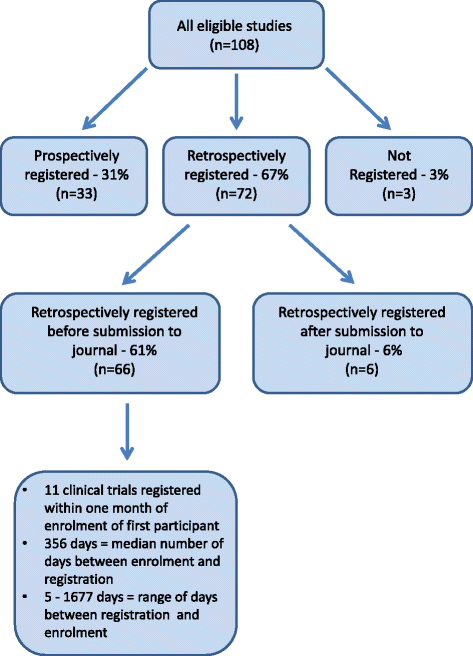
When are clinical trials registered? An analysis of prospective versus retrospective registration | Trials | Full Text

Inclusion of Patient-Reported Outcomes in Adolescent and Young Adult Phase III Therapeutic Trials: An Analysis of Cancer Clinical Trials Registered on ClinicalTrials.gov - Value in Health

Registration of Clinical Trials: Background and Implementation Ida Sim, MD, PhD Project Coordinator Department of Research Policy and Cooperation World. - ppt download

Peer reviewed evaluation of registered end-points of randomised trials (the PRE-REPORT study): protocol for a stepped-wedge, cluster-randomised trial | BMJ Open

ACT Junior Rugby Union - NEW REGISTRATION PROCESS FOR REP RUGBY ***UPDATES AT BOTTOM OF POST** The ACTJRU have now set up a New Registration process for Rep Rugby through Rugby Xplorer.

Challenges for funders in monitoring compliance with policies on clinical trials registration and reporting: analysis of funding and registry data in the UK | BMJ Open

Overview of phase IV clinical trials for postmarket drug safety surveillance: a status report from the ClinicalTrials.gov registry | BMJ Open